This fundamental chapter of NCERT Solutions for Class 10 Science Chapter 4, Carbon and its Compounds, helps us understand the remarkable versatility of carbon - the element that forms the basis of all life on Earth. From the fuel we burn for energy to the food we eat, plastics we use daily, and even the molecules that make up our bodies, carbon compounds are everywhere. This chapter is part of the comprehensive NCERT Solutions Class 1o Science series, which covers all chapters in detail.
The chapter equips students with essential skills to understand carbon's unique bonding properties, nomenclature of organic compounds, and the characteristics of important carbon compounds like hydrocarbons, alcohols, and carboxylic acids. Every solution has been designed keeping CBSE board exam patterns in mind, ensuring students develop both conceptual clarity and problem-solving confidence. By mastering this chapter, students build a strong foundation for organic chemistry and biochemistry topics in higher classes.
NCERT Solutions for Class 10 Science Chapter 4 - All Exercise Questions
Q.
Ethane, with the molecular formula C2H6 has
(a) 6 covalent bonds.
(b) 7 covalent bonds.
(c) 8 covalent bonds.
(d) 9 covalent bonds.
Q.
Butanone is a four-carbon compound with the functional group
(a) carboxylic acid.
(b) aldehyde.
(c) ketone.
(d) alcohol.
Q.
While cooking, if the bottom of the vessel is getting blackened on the outside, it means that
(a) the food is not cooked completely.
(b) the fuel is not burning completely.
(c) the fuel is wet.
(d) the fuel is burning completely.
Q.
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Q.
Draw the electron dot structures for
(a) ethanoic acid.
(b) H2S.
(c) propanone.
(d) F2.
Q.
What is a homologous series? Explain with an example.
Q.
How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
Q.
Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Q.
Why are carbon and its compounds used as fuels for most applications?
Q.
Explain the formation of scum when hard water is treated with soap.
Q.
What change will you observe if you test soap with litmus paper (red and blue)?
Q.
What is hydrogenation? What is its industrial application?
Q.
Which of the following hydrocarbons undergo addition reactions:
C2H6, C3H8, C3H6, C2H2 and CH4
Q.
Give a test that can be used to differentiate chemically between butter and cooking oil.
Q.
Explain the mechanism of the cleaning action of soaps.
Class 10 Chapter 4 Science Questions & Answers –Carbon and its Compounds
Q1. Ethane, with the molecular formula C2H6 has
(a) 6 covalent bonds.
(b) 7 covalent bonds.
(c) 8 covalent bonds.
(d) 9 covalent bonds.
Solution:
7 covalent bonds
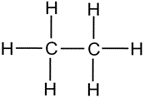
Q2. Butanone is a four-carbon compound with the functional group
(a) carboxylic acid.
(b) aldehyde.
(c) ketone.
(d) alcohol.
Solution:
Q3. While cooking, if the bottom of the vessel is getting blackened on the outside, it means that
(a) the food is not cooked completely.
(b) the fuel is not burning completely.
(c) the fuel is wet.
(d) the fuel is burning completely.
Solution:
(b) the fuel is not burning completely.
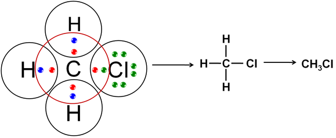
Q4. Explain the nature of the covalent bond using the bond formation in CH3Cl.
Solution:
Carbon has 4 valence electrons. It can neither lose four of its electrons nor gain four electrons as both the processes require large amount of energy. In order to complete the octet, it shares each of the four electrons with each of the three hydrogen atoms and one chlorine atom. Since, bonds are formed because of sharing of electrons, hence these are covalent bonds.
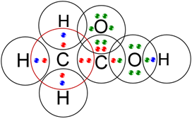
Q5. Draw the electron dot structures for
(a) ethanoic acid.
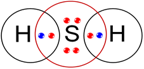
(b) H2S.
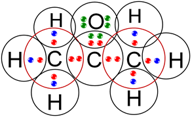
(c) propanone.
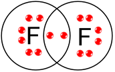
(d) F2.
Solution:
(a) Ethanoic acid
(b) H2S
(c) Propanone
(d) F2
Q6. What is a homologous series? Explain with an example.
Solution:
A homologous series is a series of organic compounds with similar general formula, possessing similar chemical properties due to the presence of the same functional group, and show a gradation in physical properties as a result of increase in molecular size and mass.
The members of homologous series are called homologue.
Compounds of same homologous series differ from their consecutive members by one carbon atom and two hydrogen atoms, i.e. by CH2.
Example:
Alkanes; such as, Methane, Ethane, Propane, Butane, etc. belong to the same homologous series.
CH4 (Methane) and C2H6 (Ethane) differ by CH2
C2H6 (Ethane) and C3H6 (Propane) differ by CH2 and so on.
Q7. How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
Solution:
Difference in Physical Properties
Ethanol is a liquid at room temperature with pleasant odour while ethanoic acid has vinegar like smell. The melting point of ethanoic acid is 17 °C. This is below room temperature and hence, it freezes during winters.
Difference in Chemical Properties
Ethanoic acid reacts with metal carbonates and metal hydrogencarbonates to form salt, water and carbon dioxide gas while ethanol does not react with them.
Example:
Ethanol does not react with sodium hydroxide (NaOH) while ethanoic acid reacts with sodium hydroxide to form sodium ethanoate and water
Ethanol is oxidised to give ethanoic acid in presence of acidified KMnO4 while no such reaction takes place with ethanoic acid in presence of acidified KMnO4.
Q8. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Solution:
Soap is a sodium or potassium salt of long-chain fatty acid which has cleansing properties in water.
Soap molecule consists of two parts hydrophobic and hydrophilic. The hydrophilic part is the ionic end of the soap molecule which is soluble in water. The hydrophobic part is the organic end and is insoluble in water. Since the dirt present in clothes is organic in nature, the hydrophobic part entraps dirt and hydrophilic part remains suspended in water. Thus, many more molecules of soap are attached to dirt having their one end suspended in water to form clusters. These clusters with entrapped dirt are known as micelle.
No, micelle will not be formed in other solvents like ethanol.
Q9. Why are carbon and its compounds used as fuels for most applications?
Solution:
The carbon and its compounds are used as fuels for most applications because
1. Most of the carbon compounds produce a lot of heat and light when burnt in air.
2. The amount of heat released can be handled and used easily.
3. Once ignited carbon and its compounds keep on burning without any additional requirement of heat energy. Saturated hydrocarbons burn with a clean flame and no smoke is produced.
4. The carbon compounds, used as fuel, have high calorific values.
Q10. Explain the formation of scum when hard water is treated with soap.
Solution:
Hard water contains Mg2+ and Ca2+ ions. Soap is sodium or potassium salt of long-chain fatty acid. When hard water is treated with soap, Mg2+ and Ca2+ ions present in hard water react with soap to form Magnesium and Calcium salts of fatty acid which are insoluble in water and are known as scum.
Q11. What change will you observe if you test soap with litmus paper (red and blue)?
Solution:
Soap is basic in nature; therefore, it will turn red litmus blue. On the other hand, the colour of blue litmus will remain blue.
Q12. What is hydrogenation? What is its industrial application?
Solution:
Hydrogenation is the process of addition of hydrogen. For example: Ethene reacts with hydrogen when heated in the presence of nickel catalyst to form ethane.
It is used for making vanaspatee ghee from vegetable oil in industries.
Q13. Which of the following hydrocarbons undergo addition reactions:
C2H6, C3H8, C3H6, C2H2 and CH4
Solution:
Unsaturated hydrocarbons(alkenes and alkynes) undergo addition reactions. In the given list, C3H6 and C2H2 are unsaturated hydrocarbons and therefore, undergo addition reactions.
Q14. Give a test that can be used to differentiate chemically between butter and cooking oil.
Solution:
Butter contains saturated fats while cooking oil contains unsaturated fats. If we add bromine water to them, the reddish brown colour of bromine will disappear in the sample of cooking oil while it will remain same in the sample of butter.
Q15. Explain the mechanism of the cleaning action of soaps.
Solution:
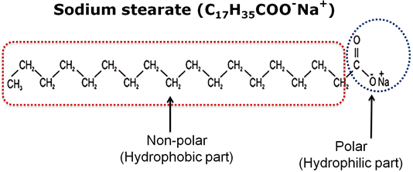
A soap molecule is a sodium or potassium salt of long chain carboxylic acid. It consists of two parts:
1. A non-polar part which consists of long chain hydrocarbons called tail. It is hydrophobic.
2. An ionic part consisting of carboxylate ion called polar head. It is hydrophilic.
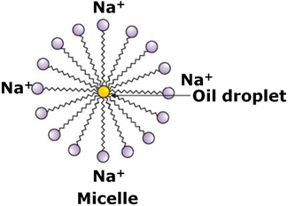
When a dirty cloth is dipped into a soap solution, its molecules arrange themselves around the dirt particles in such a way that the hydrophobic hydrocarbon chain gets embedded inside the dirt particles and the hydrophilic head projects outside the dirt particles like bristles.
As the hydrophilic part is polar, it interacts with the water molecules present around the dirt particles. This result into the formation of spherical clusters called Micelle.
The soap molecules form aggregates around the dirt particles which get pulled away from the surface of the cloth when the dirty cloth is agitated in soap solution.
The dirt particles get suspended in water due to which the soap water becomes dirty and cloth is cleaned.
More Resources of NCERT Solutions for Class 10 Science
NCERT Solutions for Class 10 Science Chapter 4 – FAQs
Q1. Why does carbon form covalent bonds and what is catenation?
Carbon forms covalent bonds because it has 4 electrons in its outermost shell and needs 4 more electrons to complete its octet. Losing or gaining 4 electrons would require enormous energy, making ionic bonding unfavorable. Instead, carbon shares electrons with other atoms (including other carbon atoms) to form stable covalent bonds. Catenation is the unique ability of carbon atoms to form long chains by bonding with other carbon atoms through strong C-C bonds. This property exists because carbon-carbon bonds are very strong and stable. Carbon can form straight chains, branched chains, and ring structures, creating millions of different compounds. This self-linking property, combined with carbon's tetravalency (ability to form four bonds), makes it the basis of all organic compounds and life itself. No other element shows catenation to the extent that carbon does.
Q2. What is the difference between saturated and unsaturated hydrocarbons?
(i)Saturated Hydrocarbons:
- These hydrocarbons only have single bonds between carbon atoms.
- Each carbon atom is bonded to as many hydrogen atoms as possible.
- Examples include Methane, Ethane, and Propane.
- Saturated hydrocarbons are less reactive and typically undergo substitution reactions.
(ii)Unsaturated Hydrocarbons:
- These hydrocarbons contain at least one double bond (alkenes) or triple bond (alkynes) between carbon atoms.
- Due to these bonds, they have fewer hydrogen atoms compared to saturated hydrocarbons.
- Examples include Ethene (alkene) and Ethyne (alkyne).
- Unsaturated hydrocarbons are more reactive and typically undergo addition reactions.
Q3. What are functional groups and why are they important in organic chemistry?
A functional group is an atom or group of atoms in a molecule that determines its characteristic chemical properties and reactions. Common functional groups include: alcohol (-OH) as in ethanol, aldehyde (-CHO) as in methanal, ketone (-CO-) as in propanone, carboxylic acid (-COOH) as in ethanoic acid, and halogens (-X) as in chloroethane. Functional groups are important because compounds with the same functional group show similar chemical properties regardless of the carbon chain length. For example, all alcohols react with sodium to produce hydrogen gas, and all carboxylic acids taste sour and turn blue litmus red. This classification simplifies the study of millions of organic compounds by grouping them into homologous series—families of compounds with the same functional group, similar chemical properties, and a gradual change in physical properties. Understanding functional groups helps predict how organic compounds will react and allows chemists to synthesize new compounds with desired properties.RetryClaude can make mistakes. Please double-check responses. Sonnet 4.5